 A key achievement of the EPAD project was the establishment of a Longitudinal Cohort Study (LCS) that has screened over 2,000 participants and collected a wide range of cognitive, clinical, neuroimaging and biomarker data to help further our understanding of the early stages of Alzheimer’s disease. EPAD has made this database open access and publicly available to the research community. On behalf of the EPAD consortium, we hope that the EPAD data will be a significant asset to the worldwide research.
A key achievement of the EPAD project was the establishment of a Longitudinal Cohort Study (LCS) that has screened over 2,000 participants and collected a wide range of cognitive, clinical, neuroimaging and biomarker data to help further our understanding of the early stages of Alzheimer’s disease. EPAD has made this database open access and publicly available to the research community. On behalf of the EPAD consortium, we hope that the EPAD data will be a significant asset to the worldwide research.
Access
All EPAD data are shared through a secure online Workspace. To access the data, you will need to make an online request…
Read More
Data
Data has been collected from participants across a wide-range of outcomes at each annual visit…
Read More
Samples access
The EPAD Bioresource consists of over 100,000 samples consisting of blood, CSF, saliva and urine…
Read More
MRI Images
This section represents an overview of the MRI data collected throughout the EPAD study…
Read More
 The LCS was initiated and the first participant screened in May 2016. Screening into the LCS was stopped on 29 February 2020 and a total of 2,096 participants had been screened. Of these 217 were screen failures and 215 have withdrawn. The number of participants that completed each visit during the study period is outlined in the infographic below.
The LCS was initiated and the first participant screened in May 2016. Screening into the LCS was stopped on 29 February 2020 and a total of 2,096 participants had been screened. Of these 217 were screen failures and 215 have withdrawn. The number of participants that completed each visit during the study period is outlined in the infographic below.
- Screened
- 2,096
- 6 month visit
- 1,596
- Year 1 visit
- 1,225
- Year 2 visit
- 421
- Year 3 visit
- 121
Although the LCS is no longer following up participants, it provides an ongoing legacy through open access to data and a vast range of possibilities for sample access. The impact of EPAD to further our understanding of Alzheimer’s disease via future research generated from global access to data and samples will be substantial.
Datasets available
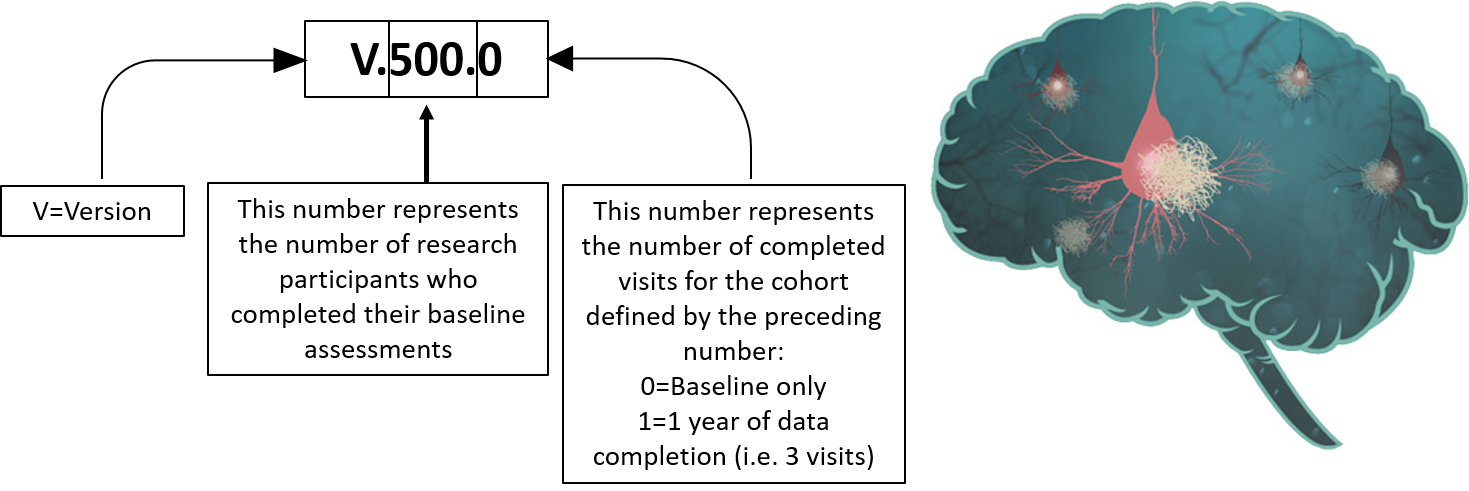
- EPAD LCS V500.0
- EPAD LCS V1500.0
- EPAD LCS V500.1
- EPAD LCS V.IMI (It represents all the data collected and processed during the IMI period of EPAD)
About the Workspaces
The analytical database for the LCS was developed and hosted by our partner Aridhia within the Digital Research Environment (DRE, formerly AnalytiXagility). Workspaces in the DRE provided the secure collaboration and data management services to integrated and distribute LCS data. The analytical database includes all of the clinical, cognitive and biomarker measurements of recruited individuals according to the EPAD LCS protocol, collected and curated according to high quality standards. The DRE was also used as part of the EPAD LCS Research Access Process (ERAP) where the DRE hosted metadata to help researchers interpret the database.
“Our data team generate the published snapshots of the analytical database, and publish snapshots and metadata. Researchers can discover the data through publications, Internet searches and go through a partially automated process to request access. A semi-automated process streamlines the review and approval of requests. Data is then usually delivered to a private Workspace in the DRE where they can download the open data, collaborate on analysis and also redeposit derived data back to the LCS.”



 A key achievement of the EPAD project was the establishment of a Longitudinal Cohort Study (LCS) that has screened over 2,000 participants and collected a wide range of cognitive, clinical, neuroimaging and biomarker data to help further our understanding of the early stages of Alzheimer’s disease. EPAD has made this database open access and publicly available to the research community. On behalf of the EPAD consortium, we hope that the EPAD data will be a significant asset to the worldwide research.
A key achievement of the EPAD project was the establishment of a Longitudinal Cohort Study (LCS) that has screened over 2,000 participants and collected a wide range of cognitive, clinical, neuroimaging and biomarker data to help further our understanding of the early stages of Alzheimer’s disease. EPAD has made this database open access and publicly available to the research community. On behalf of the EPAD consortium, we hope that the EPAD data will be a significant asset to the worldwide research. The LCS was initiated and the first participant screened in May 2016. Screening into the LCS was stopped on 29 February 2020 and a total of 2,096 participants had been screened. Of these 217 were screen failures and 215 have withdrawn. The number of participants that completed each visit during the study period is outlined in the infographic below.
The LCS was initiated and the first participant screened in May 2016. Screening into the LCS was stopped on 29 February 2020 and a total of 2,096 participants had been screened. Of these 217 were screen failures and 215 have withdrawn. The number of participants that completed each visit during the study period is outlined in the infographic below.